Our research identifies and addresses substantial and crucial gaps in the regenerative medicine field, bringing this field ever more close to clinical applications. We developed a unique decellularization procedure resulting in ECM that highly resembles human tissue, and can promote tissue regeneration without evoking an immune response (WO/2006/095342). This promising biomaterial has been the heart of diverse advanced regenerative medicine platforms that were developed in our lab in the recent years.
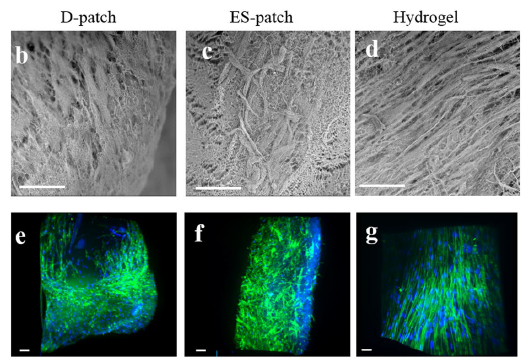
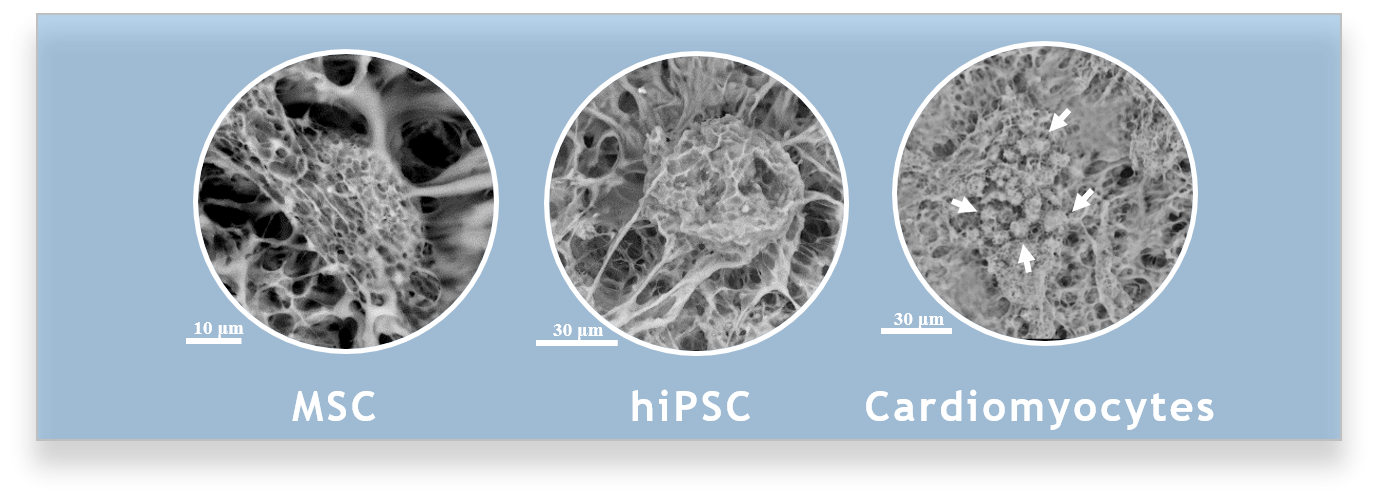
Directing the attributes of ECM-based cardiac scaffolds through processing technologies: The different cardiac scaffolds we have developed were studied for the bioeffects that the manufacturing process, modality, and 3D structure of the scaffolds may have on the scaffold’s biological outcome while using the same biomaterial. We, therefore, chose 3 different pcECM-based scaffolds, aiming to reveal their unique characteristics as well as the effects of these different characteristics on cell-scaffold interactions. While each of the scaffolds―decellularized pcECM patch, injectable pcECM hydrogel, and electrospun pcECM patch―was tested before for its own properties and proved its therapeutic potential, their unique leverages, which can guide the choice of scaffold for a particular cellular or acellular treatment were not addressed. We demonstrated that minor differences between pcECM-based scaffolds composition and micro-morphology as well as substantial differences in their mechanical properties, originating from their production process, highly affect cell-scaffolds interactions. Moreover, these effects are dependent upon the type of cells populating the scaffolds, which differently attach to them, survive, proliferate and remodel the scaffolds. We believe these revealed effects not only support the accumulating science regarding the particular benefits of each ECM-based scaffold but can also guide a more rational cardiac scaffold design for the achievement of improved cardiac regeneration.
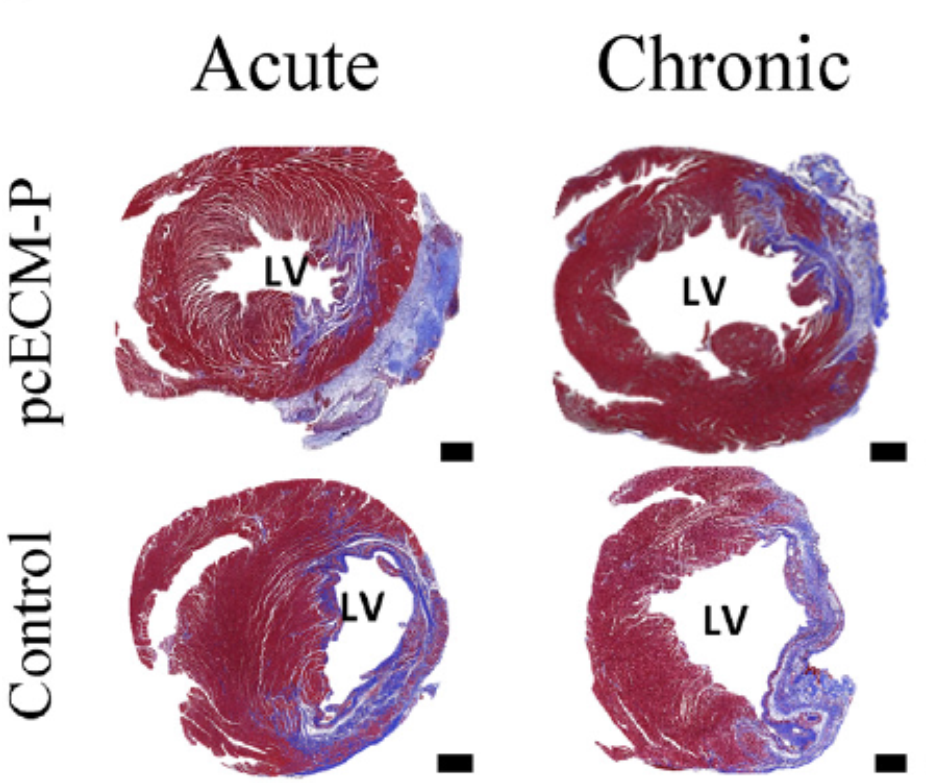
Native pECM cardiac patch scaffold: In the development of a native pECM cardiac scaffold, the decellularized cardiac pECM exhibited the desirable mechanical properties required from scaffolds used for cardiac tissue repair (1). Moreover, it was proven to be cytocompatible with fibroblasts, cardiomyocytes, and mesenchymal stem cells, which interact with the pECM and remodel it. The pECM scaffold supported cardiac cells function in vitro, thus beating in concert and expressing typical functional cardiac markers (1). In vivo, the native pECM scaffolds alone, i.e., without added biologics, were bioactive, and exerted potent efficacy, stimulating biological regenerative processes that cooperatively led to a cardiac progenitor-based restoration of function, even after scar tissue had already formed. In both acute and long-term chronic rat MI models, it promoted significant cardiac function improvement, which was correlated to progenitors and myocyte recruitment. Recruited progenitors, differentiated towards the CM lineage and were organized in a partially striated and immature muscle fiber arrangement. Concomitantly, pcECM was rapidly vascularized, and induced a constructive remodeling process (2).
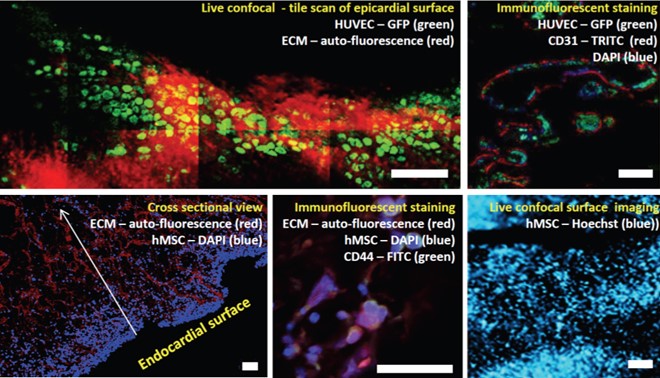
Thick vascularized porcine cardiac pECM scaffold: To enable maximal support to the infarcted heart, a full-thickness pECM scaffold was developed, which preserves the LV wall inherent vasculature. This scaffold was proved to be non-immunogenic, cell-supportive, and to preserve the native tissue ultra-structural properties (3). Through the development of a unique, custom-designed perfusion bioreactor, the capabilities of recellularizing the thick pECM scaffold were gained. Functional ex vivo angiogenesis and vascularization supported, for the first time, up to 1.7 mm thick cellularized constructs (4). A robust methodology was developed to assess the pECM maximal cell capacity, which resembled the human heart cell density, including a mathematical model predicting the co-culture dynamics of endothelial and mesenchymal stem cells (4)(5). The recellularized thick pECM was also shown to restore the micro and macro-mechanical properties of the heart tissue, thus enabling an optimal support to end-stage MI heart failure (6)(7)(8). Currently, this promising scaffold is investigated using a large-animal MI model, demonstrating very encouraging preliminary results.
Electrospun pECM cardiac patch scaffold: To harness the natural advantages of pECM in a reproducible, scalable, and controllable scaffold, the technological capabilities to electrospin whole pECM were developed (WO/2017/179042). The resulted electrospun cardiac ECM scaffold preserved the composition of ECM, self-assembled into the same microstructure of cardiac ECM, and, above all, preserved key cardiac mechanical properties. It supported cell growth and function, and demonstrated biocompatibility in vitro and in vivo. An ongoing research currently addresses the efficacy of this novel scaffold in a chronic MI rat model, thus revealing its great regenerative potential (9).
Injectable pECM cardiac scaffold: To allow a minimally invasive cardiac regenerative treatment, injectable pECM-based hydrogels were developed. Through the addition of a natural crosslinker and a biopolymer, better control of the gel’s mechanical properties was achieved, while still leveraging the ECM biological activity. The pECM-based hydrogels were cell supportive and biocompatible. Most importantly, their application in treating acute and long-term chronic MI in rat demonstrated a significant improvement in cardiac dimensions and function eight weeks post-treatment (10). Currently, large-animal studies are conducted to reveal the full potential of the pECM-based hydrogel as a stand-alone cardiac scaffold or as a cell-delivery platform. Concomitantly, the mechano-biological interplay between cells and the pECM hydrogels is thoroughly investigated, aiming to produce a wide scientific basis for the pECM tissue-specific properties and bioactivity.
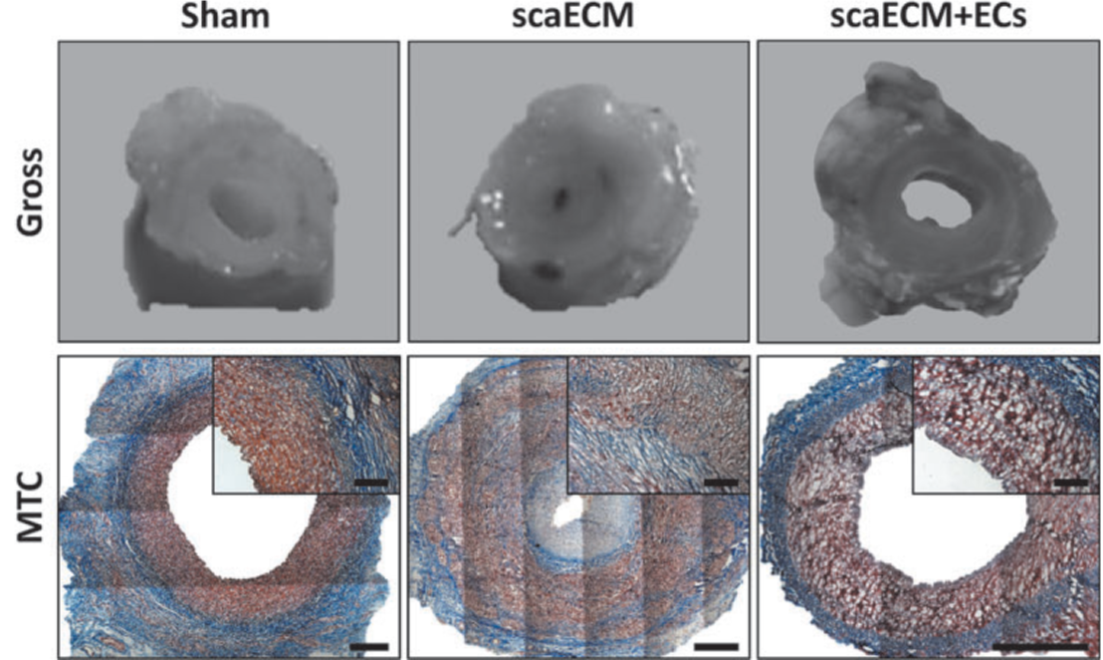
Vascular pECM bio-graft: In the field of arterial regenerative medicine, a pECM engineered vascular graft for arterial bypassing was developed. This pECM vascular graft, produced by enzymatic and detergent decellularization of small caliber arteries, preserved major extracellular matrix proteins and the mechanical properties of the native artery. The dynamic co-culturing of smooth muscle cells (SMC) and endothelial cells was achieved with the design of a costume-made dynamic perfusion bioreactor that led to infiltration, migration, and proliferation of SMC toward the media and to a confluent endothelium formation on the luminal surface (11). In vivo, endothelialized pECM vascular grafts were transplanted as an interposition carotid artery graft in a porcine model. Following transplantation, dynamically endothelialized grafts remained patent for 6 weeks, provided excellent antithrombogenic activity, inhibited intimal hyperplasia formation, and encouraged media wall infiltration and reorganization with recruited host SMCs (12).