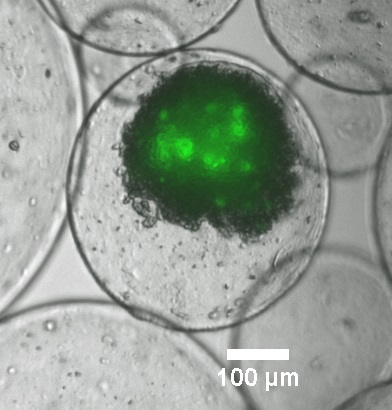
Cell microencapsulation is a fundamental technique in cell-based therapy allowing continuous delivery of biologic drugs. In the prestigious journal, Nature Biotechnology (2001), we reported an alginate-encapsulation cells system, designed to deliver anti-angiogenic biologic drugs, which led to significant inhibition of glioblastoma tumors. Since then, we mainly focus on the development of new encapsulation platforms which will facilitate the prospective clinical application of this cell-based therapy approach. These include polymeric system comprising alginate and chitosan, exhibiting superior mechanical properties and biocompatibility a system which releases anti-inflammatory drugs along with the therapeutic protein and a system for encapsulation of transfected mesenchymal stem cells secreting antiangiogenic proteins for the treatment of solid tumors. In addition, we developed a process to create microcapsules from solubilized decellularized extracellular matrix (ECM), from different origins. These microcapsules provide the cells with a more natural and supportive microenvironment than conventional alginate microcapsules, which dramatically improve cell function.
Mesenchymal stem cells entrapped in microcapsules made of pancreatic extracellular matrix:
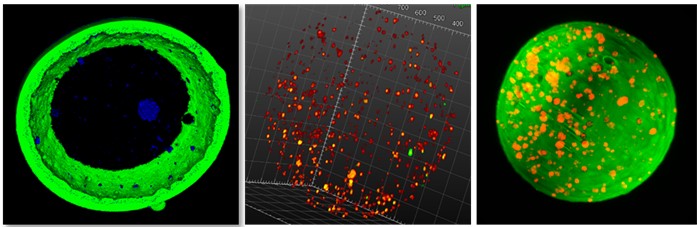
Pancreatic pECM microscaffolds: For the development of an artificial pancreas platform for the treatment of diabetes, a novel microencapsulation technology was established that is based on solubilized whole pancreatic pECM (WO/2014/037942A1). The pECM microcapsules were used to entrap human liver cells and mesenchymal stem cells that were induced to differentiate into glucose-regulated insulin-producing cells. This work demonstrated that the pECM-microcapsule platform provided a natural fibrous 3D niche, supporting cell viability and differentiation, while significantly improving insulin delivery. In vivo, pECM-encapsulated cells were shown to be non-immunogenic, and most importantly, to significantly improve the glycemic control in diabetic mouse preclinical model, thus establishing a proof-of-concept for this new microscaffolds-based insulin delivery platform (13). An ongoing research aims to establish a pECM-based encapsulation platform for the entrapment of pancreatic islets, thus harnessing their natural glucose-regulated insulin secretion in an immunoisolated biosupportive platform for diabetes therapy.