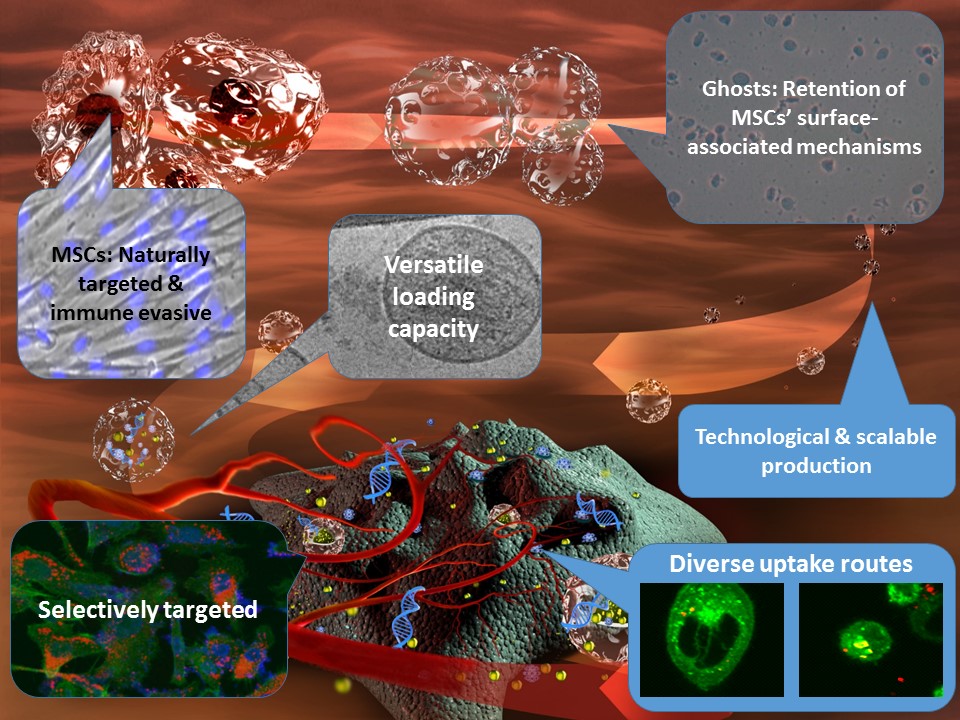
Nano-ghosts technology offers a novel versatile and highly selective targeted delivery platform, which, owing to its natural origins and scalable technological derivation, may overcome the limitations of conventional delivery systems. This platform is based on unique cell-derived nano-vesicles, termed nano-ghosts (NGs), produced from the cell membranes of naturally targeted mesenchymal stem cells (MSCs). MSC-derived NGs (MSC-NG) are produced by a scalable and reproducible process consisting of hypotonic treatment and mild homogenization—which removes the cell cytoplasm to attain the ghost cells—followed by size reduction to produce the NGs. Similar to synthetic liposomes, the NGs may be loaded with any drug or therapeutic of choice during their size reduction or post preparation by active means (e.g., electroporation). On top of retaining MSCs unique targeting properties, the NGs also benefit from the surface associated capabilities that make allogeneic MSC immune-evasive, biocompatible, and highly tolerated, even in multiple administrations.
Nano-Ghosts Executive Summary [PDF]
NANO-GHOSTS MANUFACTURING & OUTLOOK
NANO-GHOSTS TECHNOLOGY OVERVIEW
SELECTED PUBLICATIONS:
Nanoghosts derived from mesenchymal stem cells and retaining their unique surface-associated tumor-targeting capabilities were redesigned as a selective and safe universal nonviral gene-therapy platform. pDNA-loaded nanoghosts efficiently targeted and transfected diverse cancer cells, in vitro and in vivo, in subcutaneous and metastatic orthotopic tumor models, leading to no adverse effects. Nanoghosts loaded with pDNA encoding for a cancer-toxic gene inhibited the growth of metastatic orthotopic lung cancer and subcutaneous prostate cancer models and dramatically prolonged the animals’ survival.
The ultimate goal in cancer therapy is achieving selective targeting of cancer cells. We report a novel delivery platform, based on nanoghosts (NGs) produced from the membranes of mesenchymal stem cells (MSCs). Encompassing MSC surface molecules, the MSC-NGs retained MSC-specific in vitro and in vivo tumor targeting capabilities and were cleared from blood-filtering organs. MSC-NGs were found to be biocompatible. Systemic administration of drug loaded MSC-NGs demonstrated 80% inhibition of human prostate cancer.
Liposomal compositions and uses of same. Patent App. WO 2011024172 A3, CN102596179A, EP2470164A2, US20120164214.
Compositions comprising liposomes composed of whole cell membrane fraction are provided. The liposomes may be attached to, or encapsulate a pharmaceutical agent. Also provided are methods of generating and using these liposomes.